In 2022, the Food and Drug Administration (FDA) approved 37 new drugs and therapies. Prior to being studied in humans, drugs undergo preclinical testing in animals to ensure they are safe. Drugs that are found to be safe in animals may then be tested in humans for safety and effectiveness during the first three phases of clinical research required by the FDA for drug approval. After the FDA approval, phase four studies may be required to monitor the long-term effects of treatments in the market.
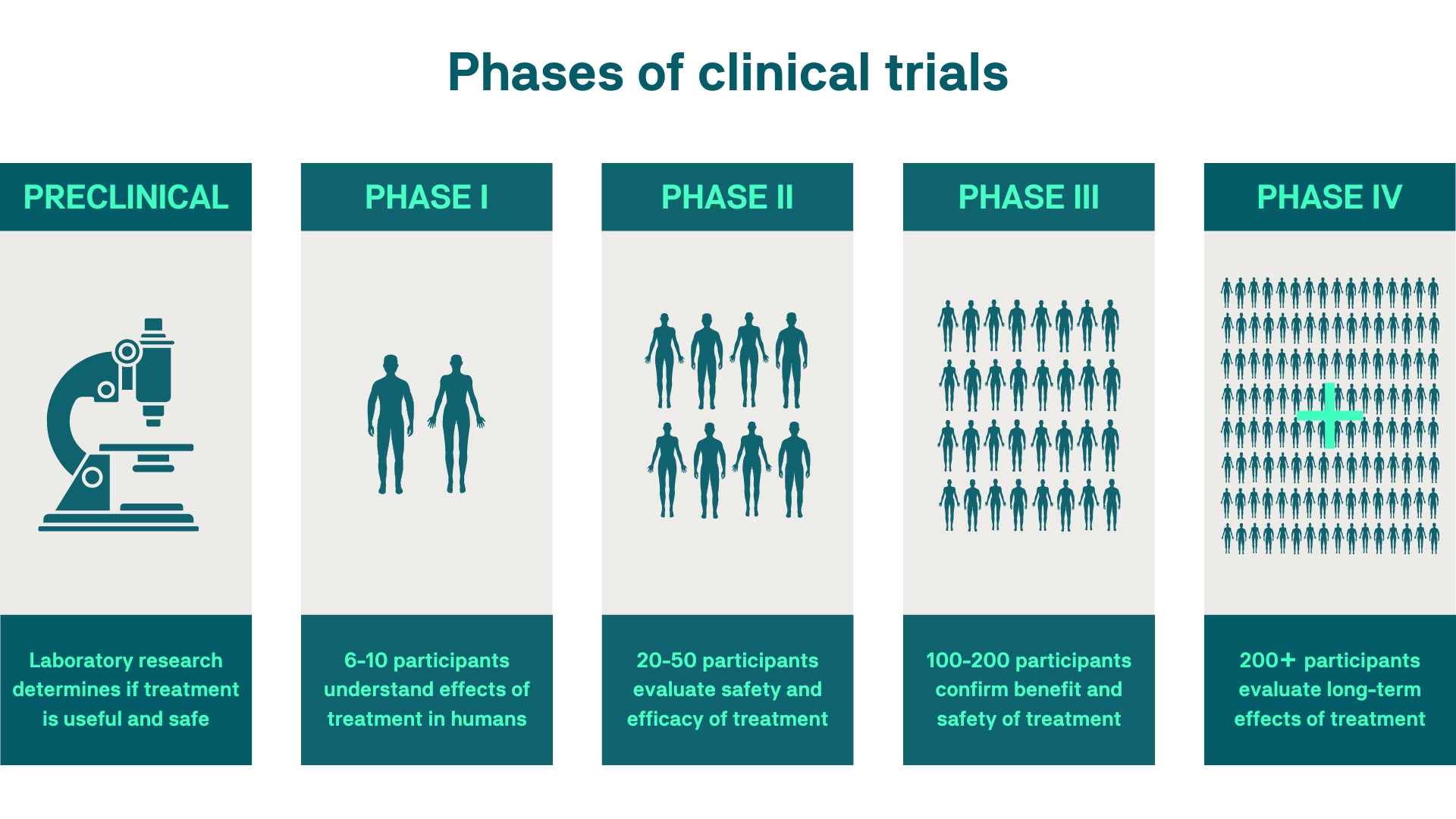
Image source: https://gene.vision/clinical-trials/
“Clinical trials are essential to finding new treatments and ways of preventing and treating diseases,” said Dr. Glen Stettin, senior vice president and chief innovation officer at Evernorth, the health services division of The Cigna Group. “Clinical trials are important because they allow researchers to understand whether new treatments are safe and effective, and how new treatments compare to existing treatments. Clinical trials help drug companies bring new medicines to market. These medicines may treat previously untreatable conditions or represent safer and more effective new treatments for people with conditions that needed better options.”
At Evernorth, we have a team working across our organization to help connect patients with relevant clinical trials. This team includes clinicians, data scientists who help match patients to trials and project managers with background experience in research and clinical trials. This team is committed to making clinical trials available as an option for care during relevant touchpoints that Evernorth has with patients.
We sat down with Ria Westergaard, director of product innovation and a pharmacist who leads the Clinical Trials team, to better understand the work they do and how they are helping improve patient outcomes.
Talk to me about your role at Evernorth.
I worked in retail pharmacy for eight years prior to joining Evernorth. I saw firsthand the challenges patients, physicians, and pharmacies faced daily due to misunderstandings about pharmacy benefit and insurance coverage, and especially coverage for experimental treatments, which most plans do not cover.
Working for Evernorth has allowed me to influence health at the population level, as I started my career here as a clinician to design formularies and coverage policies. Starting in 2017, I transitioned into my current role. I lead our clinical trial solutions, a program that’s been active since 2011.
What drives me every day is introducing clinical research studies to as many people as possible. The term “clinical trials” brings up a lot of myths and misconceptions. For example, many people think that clinical trials are only for people who have exhausted all other treatment options. But the reality is that a spectrum of clinical trials are available for all types of individuals, not just those who have tried all currently approved treatments for their condition. My team and I are on a mission to bust these types of myths, advance health care, and improve health equity by increasing access to research.
Talk to me about the Clinical Trials team and the work you do.
Our team has been tasked with embedding clinical trials throughout the larger organization. We help studies’ sponsors and the organizations that support them to recruit people into clinical trials faster. To date, we’ve introduced clinical trials to 200,000 people, across more than 40 studies, in which approximately 10,000 people have chosen to participate.

Some members of the Clinical Trials team at an industry event earlier this year.
Each day, the Cigna and Evernorth brands have thousands of interactions with patients. We can leverage those touchpoints to educate patients about clinical trials, the benefits of participation, and offer to connect patients to relevant trials. For study planning, we layer on social determinants of health (SDOH) data to ensure that trials represent the diversity of the general population. Study sponsors also use this information when planning studies for improved engagement and retention of participants throughout the study by ensuring the study staff address social challenges. And finally, the team also identifies and engages providers who may be treating high volumes of potential participants.
We support all types of research studies, phase two through phase four, always keeping the patient’s best interest in mind. More than 24,000 studies are actively recruiting in the United States right now, and 85% of them will not recruit enough patients in time. Our goal is to help advance research and bring new treatments to the market faster by increasing awareness of these study opportunities. Some studies are survey-based, and a person can participate one time, from home, or perhaps use an app over time. These types of virtual studies gained traction during COVID lockdowns. Other studies are testing a new medication and might require visiting a clinic or research center.
Clinical trials are reviewed and approved by an independent institutional review board to ensure the study is ethical. In addition, each study we offer to Evernorth members is evaluated by our clinicians, leadership, and legal privacy counsel.
Can you share some details on a clinical trial the team recruited for and what the outcome was?
A recent study we recruited for focused on men’s health, and we contacted those who had filled a prescription for a specific medication with an invitation to participate. The study sponsor had a goal to recruit 150 men. For that study, 75% of the individuals who expressed interest in participating passed further screening and enrolled. We were able to reach the enrollment goals for this clinical study in just 19 days.
The typical way that study sponsors recruit is through social media and search marketing, and the average click-throughs tend to be very low — under 1%. When we communicate directly to people, we’ve already identified who could fit a study, and as a result we see a five times higher response rate on average. Two-thirds of those who respond will pass the screening and enroll in the study. This is exciting work because it’s providing an important public service and it’s the right thing to do. Patients value learning about cutting edge research that can help them or people like them.
What’s in store for the future? What do you see as some of the biggest opportunities?
We see a big opportunity within the oncology space. The National Comprehensive Cancer Network (NCCN), the organization that develops cancer treatment guidelines, states in every guideline: “NCCN believes that the best place for any patient with cancer is in a clinical trial,” but only 6% of patients are participating. The reason is simple: they don’t know about the clinical trials that could be a good fit for them. There is so much innovation that is happening in cancer treatment, and so the team is going to be putting a big focus on building awareness and participation in cancer-related clinical trials.