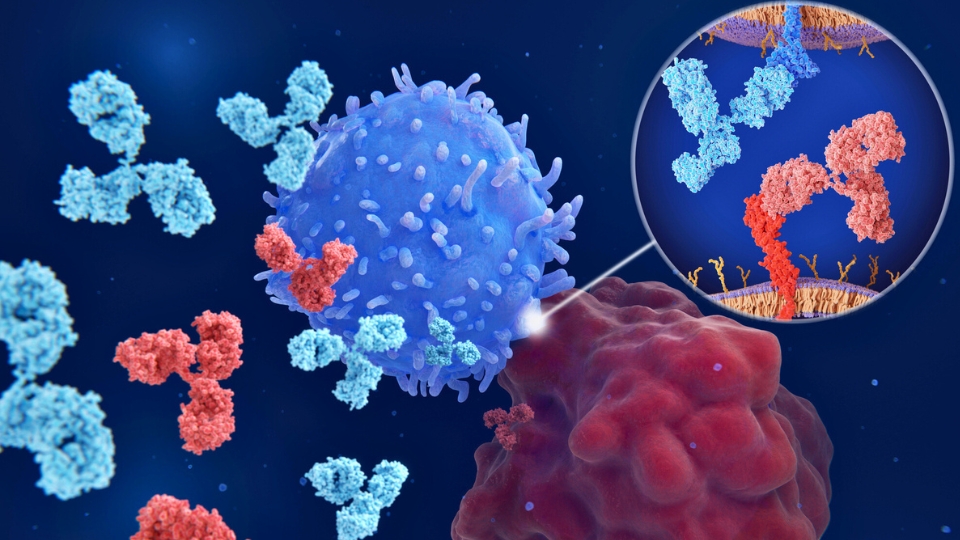
Cancer care has taken a significant step forward with the introduction of a new class of medications, immune checkpoint inhibitors (ICIs), which enable the patient’s immune cells to destroy cancer cells.
The value of immune checkpoint inhibitors
Since the first ICIs were approved by the U.S. Food and Drug Administration (FDA), survival rates for melanoma, lung cancer, and kidney cancer have improved greatly. At the same time, the amount spent on these medications has soared — from $360 million globally in 2011, the year they launched, to an estimated $37.3 billion in 2022. Those costs will continue to rise by an estimated 19% annually through 2030.
“The introduction of ICIs has been a game changer in cancer treatments,” said Dr. Roseleen Charania, Associate Chief Medical Officer for oncology programs at Evernorth, the health services division of The Cigna Group. “When ICIs are prescribed and administered in an optimal manner, they can provide significant benefits, markedly improving survival rates in comparison to other therapies.”
By 2021, 12.1% of oncology patients were receiving ICIs, a 47.5% increase since 2018 that was driven primarily by expanded use in treating breast, colorectal, and prostate cancer.
In a report, the Evernorth Research Institute looked at how ICIs and other cancer medicines are being utilized today, analyzing utilization and cost data from 47.8 million people, along with examining the data of a smaller group who were treated with infusions of pembrolizumab, the ICI most commonly used for metastatic lung cancer.
Read on for the three key actionable insights our research uncovered that can help health plans optimize cancer care.
Insight 1: The infusion setting for ICI treatment drives costs
Almost two-thirds of ICI treatments are administered in a hospital’s outpatient clinic, where the cost of care averages $157,000 per patient annually, compared with an average cost of $87,000 for treatments administered in a doctor’s office.
“The cost difference is associated with a far higher drug cost for these patients,” said Urvashi Patel, Ph.D., vice president of the Evernorth Research Institute. “Our research didn’t see any differences in the number of infusions or the size of the doses. This represents an opportunity to control spending on these important treatments without affecting the quality of care.”
Despite that cost discrepancy, the percentage of infusions in hospital outpatient settings continues to rise, with almost two of every three patients receiving ICI treatments in a hospital setting in 2021. In part, this is due to the large number of oncology practices that have been acquired in recent years by hospitals and venture capital firms. By the start of 2022, hospitals owned more than one in four physician practices.
Insight 2: Health disparities and genomic testing greatly impact ICI therapy
Not all patients are appropriate candidates for ICI therapy. To identify those who are, genomic testing for key biomarkers is crucial. Yet our research found these best practices are not always followed. For example, our research found that 30% of those treated with pembrolizumab, the medication commonly used for metastatic lung cancer, did not undergo prior biomarker testing.
Patients who live in areas with high social needs, such as lower incomes, lower average educational attainment, and limited transportation, were even less likely to have undergone genomic testing, with only 24% receiving prior biomarker testing.
Additionally, the research found that people who had not been tested were more likely to discontinue ICI therapy: 53% of patients who had not undergone genomic testing stopped receiving treatment after the fifth treatment, compared with 44% of patients who had been treated.
Insight 3: Applying evidence-based guidelines can help prevent wasteful spending in ICI treatment
ICI treatment for advanced stage non-small cell lung cancer should last 24 months or less, according to the FDA labeling and the National Comprehensive Cancer Network, and a recent study found no evidence to suggest that indefinite immunotherapy improves the patient’s survival. However, Evernorth found that 12.2% of patients being treated with pembrolizumab for metastatic lung cancer received infusions of the medication for 24 to 30 months, 5.3% were treated from 30 to 36 months, and 4.6% received treatment for more than 36 months.
That data identifies an opportunity to reduce waste by stopping treatment at an early but appropriate time, the study concludes. It also sees an opportunity to reduce waste by utilizing supportive care therapy instead of aggressive treatments for patients entering end-of-life scenarios, for example those in hospice. These alternative options can increase quality of life, making their last days more comfortable while minimizing financial hardships.
“As the use of ICIs continues to grow, so will their cost impact on health plans,” Patel said. “We designed this report to help plans address current trends in oncology, and we hope these findings will enable more appropriate usage of ICIs, improve patient outcomes, and reduce waste.”
Originally published on 7/14/2023 and updated on 3/27/2025.